Medical Therapy
Medications usually constitute the first-line treatments for hair loss. The purpose of medical therapy is to stabilise the condition and, in some cases, promote hair regrowth.
Dr Bhoyrul is experienced in the medical management of non-scarring and scarring forms of alopecia in adults and children. He adopts an evidence-based approach and delivers personalised treatment according to the nature and severity of the condition. He strives to integrate patients' values, goals and expectations into treatment decision-making.
Dr Bhoyrul is experienced in the use of several agents to treat scalp and hair disorders, including:
- Topical, oral and sublingual minoxidil
- 5α-reductase inhibitors e.g., finasteride and dutasteride
- Anti-androgen therapy e.g., spironolactone and bicalutamide
- Topical and oral antibiotics e.g., clindamycin and doxycycline
- Topical, intralesional and oral corticosteroids
- Oral isotretinoin
- Systemic immunosuppressants e.g., methotrexate and ciclosporin
- Topical and oral Janus kinase inhibitors e.g., tofacitinib and baricitinib
- Biologics e.g., adalimumab
Low-Dose Oral Minoxidil
Minoxidil is a potent arteriolar vasodilator which was approved by the US Food and Drug Administration (FDA) for the treatment of severe refractory hypertension in 1979. Oral minoxidil has dose-dependent and predictable side effects at doses between 10-40 mg daily. The most significant systemic side effects with these doses are postural hypotension, fluid retention, tachycardia, pericarditis and nausea. However, the most common adverse effect of oral minoxidil is hypertrichosis, which occurs in approximately 80% of patients. Based on this serendipitous adverse effect, minoxidil was repurposed for the treatment of androgenetic alopecia, and the topical formulation was developed in 1987 for the treatment of male and female pattern hair loss.
The precise mechanism whereby minoxidil promotes hair growth is not entirely clear. Minoxidil sulphate is the biologically active metabolite of minoxidil which induces hair growth and is also responsible for the haemodynamic side effects. Minoxidil sulphate shortens the telogen phase of the hair growth cycle, thus causing premature transition to anagen. It also prolongs anagen, resulting in increased hair length and diameter. The initial hair growth-promoting effects of minoxidil occur after approximately two months, with maximal effects observed at four months.
The use of low-dose oral minoxidil (LDOM, 0.25-5 mg) to treat hair loss was first reported by Prof Rodney Sinclair, Dr Bhoyrul’s mentor and colleague in Melbourne. Low-dose oral minoxidil is increasingly preferred over topical minoxidil for the treatment of various forms of alopecia for the following reasons: (1) many patients find oral administration more convenient that the topical application of a lotion or foam, (2) topical application is operator-dependent i.e., some parts of the scalp may be missed in those with diffuse alopecia, (3) LDOM circumvents the local side effects associated with topical minoxidil such as irritant and allergic contact dermatitis, and (4) the percutaneous absorption of topical minoxidil is only 1.5-4%; terminal anagen hair bulbs, which are the site of action of minoxidil, reside deep in the skin (at the level of the subcutis) and are largely inaccessible to topical therapies.
There is now ample evidence to show that LDOM is effective for the treatment of male and female pattern hair loss. In addition, LDOM has been shown to improve hair density in patients with traction alopecia, loose anagen hair syndrome and lichen planopilaris.
Sublingual Minoxidil
Oral minoxidil is at least 90% absorbed from the gastrointestinal tract and is metabolised extensively in the liver by the hepatic sulfotransferase enzyme (SULT2A1) to minoxidil sulphate, which is biologically active. Minoxidil sulphate lowers blood pressure by opening adenosine triphosphate-sensitive potassium channels in vascular smooth muscle cells, and is therefore responsible for the haemodynamic side effects of postural hypotension and tachycardia.
Sublingual minoxidil is absorbed directly into the bloodstream and therefore bypasses hepatic first-pass metabolism. It is converted by follicular sulfotransferase (SULT1A1) to minoxidil sulphate. Therefore, the bioavailability of minoxidil sulphate in the hair follicle is greater with sublingual minoxidil. Furthermore, circulating minoxidil is biologically inactive, so the risk of haemodynamic side effects is lower with sublingual minoxidil.
The use of sublingual minoxidil to treat hair loss was also first reported by Prof Rodney Sinclair in the Journal of the European Academy of Dermatology and Venereology. Dr Bhoyrul and his colleagues have had success treating hair loss with sublingual minoxidil.
Bicalutamide
Bicalutamide is a non-steroidal anti-androgen which was approved by the FDA for the treatment of prostate cancer in 1995. The usual dose of bicalutamide for this indication is 150 mg. There is now data to suggest that bicalutamide at lower doses (10-50 mg) can improve hair density in women with female pattern hair loss.
A study of 308 women with female pattern hair loss treated with bicalutamide either in monotherapy or in combination with other drugs (LDOM or spironolactone) by Dr Bhoyrul and his colleagues resulted in an improvement in hair thickness. Adverse effects were uncommon.
Given its mechanism of action, bicalutamide may be a good therapeutic option for women with hair loss due to androgen excess as well as women with hirsutism.
Janus Kinase Inhibitors
Janus kinase (JAK) inhibitors e.g., tofacitinib, baricitinib, ruxolitinib, upadacitinib and abrocitinib belong to a class of immunomodulatory drugs which are used for the treatment of inflammatory conditions such as atopic dermatitis, psoriatic arthritis, rheumatoid arthritis, systemic lupus erythematosus and myelodysplastic disorders.
The Janus kinase-signal transducer and activator of transcription (JAK-STAT) pathway plays a pivotal role in the pathogenesis of alopecia areata. Unrestrained activation of the JAK/STAT pathways contributes to a number of autoimmune diseases, including alopecia areata. Pharmacologic inhibition of the JAK-STAT pathway with JAK inhibitors has been shown to induce hair regrowth in patients with alopecia areata in several uncontrolled studies. Following the findings of a randomised controlled trial published in the New England Journal of Medicine, the selective JAK1 and JAK2 inhibitor baricitinib was FDA-approved for the treatment of alopecia areata in adults.
Dr Bhoyrul is experienced in the use of topical and oral JAK inhibitors, and has co-authored publications on the use of these drugs in patients with alopecia areata as well as certain types of scarring alopecia.
Medical Therapy
Dr Bhoyrul is experienced in the medical management of non-scarring and scarring alopecias. He adopts an evidence-based approach and will tailor therapy according to the condition(s), co- morbidities and patient preference. Dr Bhoyrul is experienced with the use of the following agents to treat hair loss conditions:
- Topical, oral and sublingual minoxidil
- 5α-reductase inhibitors e.g., finasteride and dutasteride
- Anti-androgen therapy e.g., spironolactone and bicalutamide
- Topical and oral antibiotics e.g., clindamycin and doxycycline
- Topical, intralesional and oral corticosteroids
- Oral isotretinoin
- Systemic immunosuppressants e.g., methotrexate and ciclosporin
- Topical and oral Janus kinase inhibitors e.g., tofacitinib and baricitinib
- Biologics e.g., adalimumab
Low-Dose Oral Minoxidil
Minoxidil is a potent arteriolar vasodilator which was approved by the US Food and Drug Administration (FDA) for the treatment of severe refractory hypertension in 1979. Oral minoxidil has dose-dependent and predictable side effects at doses between 10-40 mg daily. The most significant systemic side effects with these doses are postural hypotension, fluid retention, tachycardia, pericarditis and nausea. However, the most common adverse effect of oral minoxidil is hypertrichosis, which occurs in approximately 80% of patients. Based on this serendipitous adverse effect, minoxidil was repurposed for the treatment of androgenetic alopecia, and the topical formulation was developed in 1987 for the treatment of male and female pattern hair loss.
The precise mechanism whereby minoxidil promotes hair growth is not entirely clear. Minoxidil sulphate is the biologically active metabolite of minoxidil which induces hair growth and is also responsible for the haemodynamic side effects. Minoxidil sulphate shortens the telogen phase of the hair growth cycle, thus causing premature transition to anagen. It also prolongs anagen, resulting in increased hair length and diameter. The initial hair growth-promoting effects of minoxidil occur after approximately two months, with maximal effects observed at four months.
The use of low-dose oral minoxidil (LDOM, 0.25-5 mg) to treat hair loss was first reported by
Professor Rodney Sinclair, Dr Bhoyrul’s mentor and colleague in Melbourne. LDOM is increasingly
preferred over topical minoxidil for the treatment of various forms of alopecia for the following
reasons: (1) many patients find oral administration more convenient that the topical application of a
lotion or foam, (2) topical application is operator-dependent i.e., some parts of the scalp may be
missed in those with diffuse alopecia, (3) LDOM circumvents the local side effects associated with
topical minoxidil such as irritant and allergic contact dermatitis, and (4) the percutaneous absorption
of topical minoxidil is only 1.5-4%; terminal anagen hair bulbs, which are the site of action of
minoxidil, reisde deep in the skin (at the level of the subcutis) and are largely inaccessible to topical
therapies.
There is now ample evidence to show that LDOM is effective for the treatment of male and female
pattern hair loss. In addition, LDOM has been shown to improve hair density in patients with traction
alopecia, loose anagen hair syndrome and lichen planopilaris.
Sublingual Minoxidil
Oral minoxidil is at least 90% absorbed from the gastrointestinal tract and is metabolised extensively in the liver by the hepatic sulfotransferase enzyme (SULT2A1) to minoxidil sulphate, which is biologically active. Minoxidil sulphate lowers blood pressure by opening adenosine triphosphate- sensitive potassium channels in vascular smooth muscle cells, and is therefore responsible for the haemodynamic side effects of postural hypotension and tachycardia.Medications are available to treat pattern (hereditary) baldness. The most common options include:
Sublingual minoxidil is absorbed directly into the bloodstream and therefore bypasses hepatic first-
pass metabolism. It is converted by follicular sulfotransferase (SULT1A1) to minoxidil sulphate.
Therefore, the bioavailability of minoxidil sulphate in the hair follicle is greater with sublingual
minoxidil. Furthermore, circulating minoxidil is biologically inactive, so the risk of haemodynamic
side effects is lower with sublingual minoxidil.
The use of sublingual minoxidil to treat hair loss was also first reported by Professor Rodney Sinclair
in the Journal of the European Academy of Dermatology and Venereology. Dr Bhoyrul and his
colleagues have had success treating hair loss with sublingual minoxidil.
Bicalutamide

Bicalutamide is a non-steroidal anti-androgen which was approved by the FDA for the treatment of prostate cancer in 1995. The usual dose of bicalutamide for this indication is 150 mg. There is now data to suggest that bicalutamide at lower doses (10-50 mg) can improve hair density in women with female pattern hair loss.
A study of 308 women with female pattern hair loss treated by our group with bicalutamide either in
monotherapy or in combination with other drugs (LDOM or spironolactone) resulted in an
improvement in hair thickness. Adverse effects were uncommon.
Given its mechanism of action, bicalutamide may be a good therapeutic option for women
with hair loss due to androgen excess as well women with hirsutism.
Janus Kinase Inhibitors

Janus kinase (JAK) inhibitors e.g., tofacitinib, baricitinib and ruxolitinib belong to a class of immunomodulatory drugs which are used for the treatment of inflammatory conditions such as atopic dermatitis, psoriasitic arthritis, rheumatoid arthritis, systemic lupus erythematosus and myelodysplastic disorders.
The Janus kinase-signal transducer and activator of transcription (JAK-STAT) pathway plays a pivotal role in the pathogenesis of alopecia areata. Unrestrained activation of the JAK/STAT pathways contributes to a number of autoimmune diseases, including alopecia areata. Pharmacologic inhibition of the JAK-STAT pathway with JAK inhibitors has been shown to induce hair regrowth in patients with alopecia areata in several uncontrolled studies. Following the findings of a randomised controlled trial published in the New England Journal of Medicine, the selective JAK1 and JAK2 inhibitor baricitinib was now FDA-approved for the treatment of alopecia areata in adults. Dr Bhoyrul is experienced in the use of topical and oral JAK inhibitors, and has co- authored publications on the use of these drugs in patients with alopecia areata as well as certain types of scarring alopecia.
Acute Telogen Effluvium

DS R Temple Before

DS R Temple After
Alopecia Areata

AD Before

AD After

AH Before

AH After

AI Eyebrows Before

AI Eyebrows After

Bari After Top GC

Bari After Back GC

Bari Before Back GC

Bari Before Top GC

EC Before 2

EC After 2

HC After

HC Before

JD After

JD Before
DLE

DLE Before

DLE After
FFA

FFA Before

FFA After
FPHL

Crea Vertex After

Crea Vertex Before

DL After Crown

DL After Midfrontal

DL Before Crown

DL Before Midfrontal

DP After

DP Before

FL After

FL Before

KV After

KV Before

PH After

PH Before
MPHL

AA After
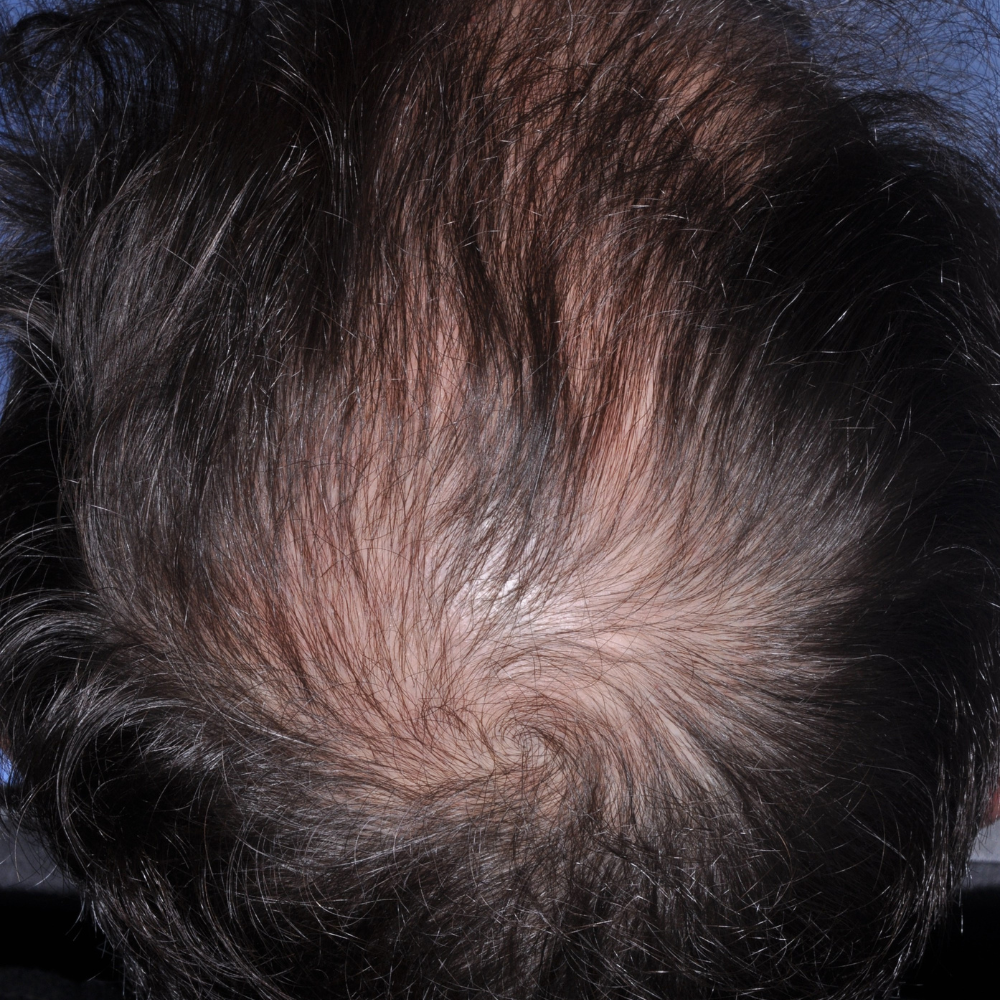
AA Before

AL After
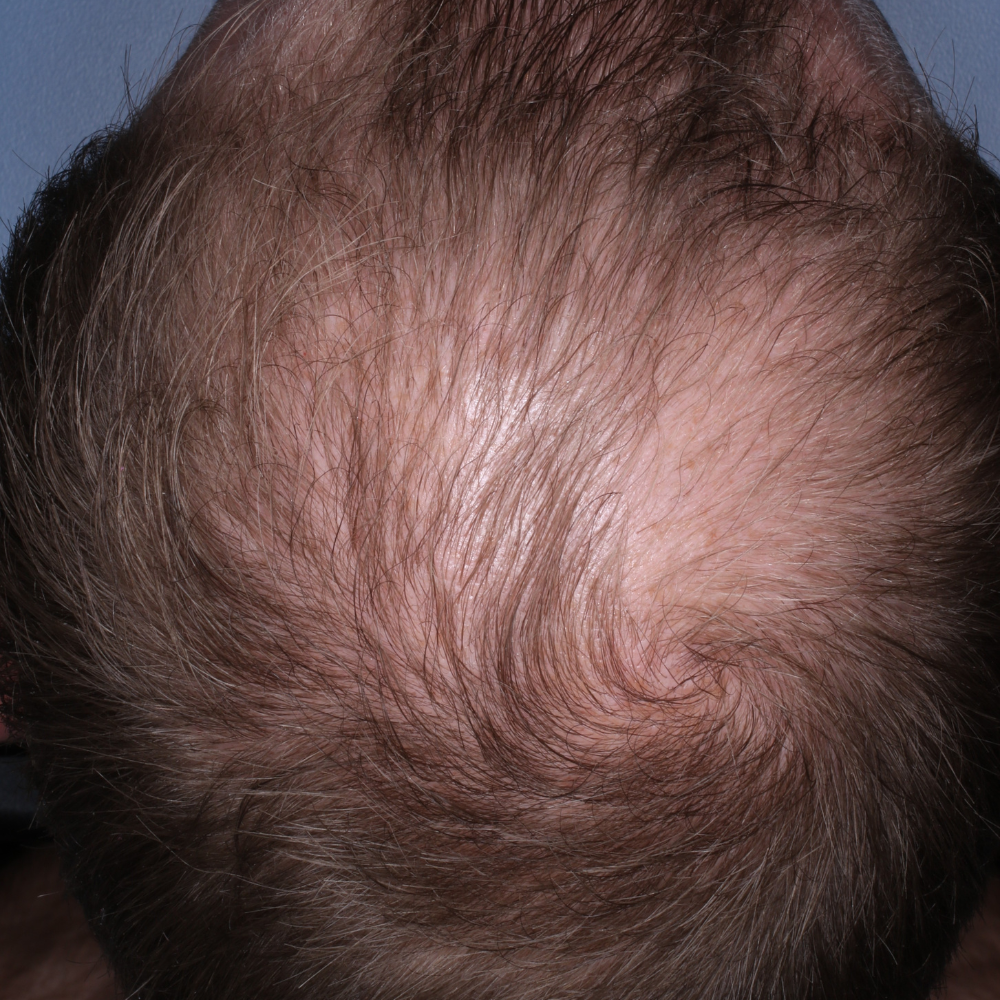
AL Before
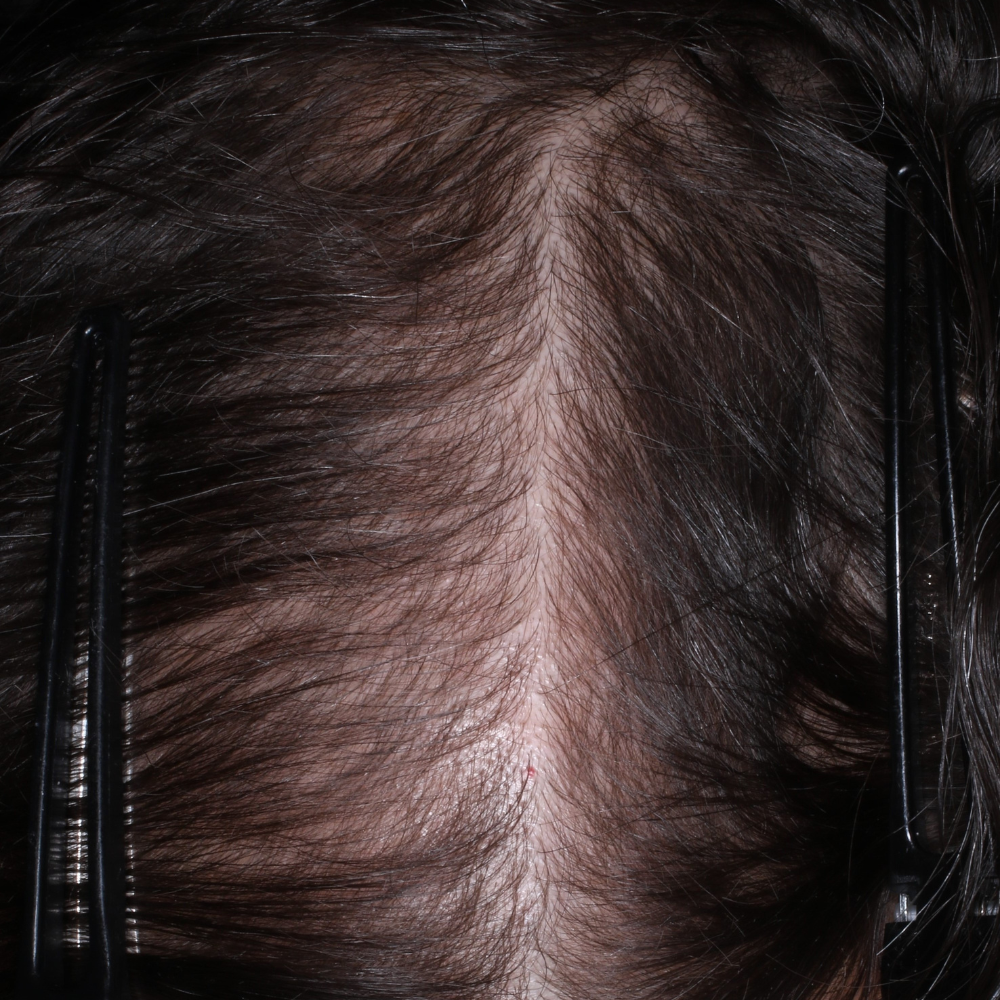
BB After
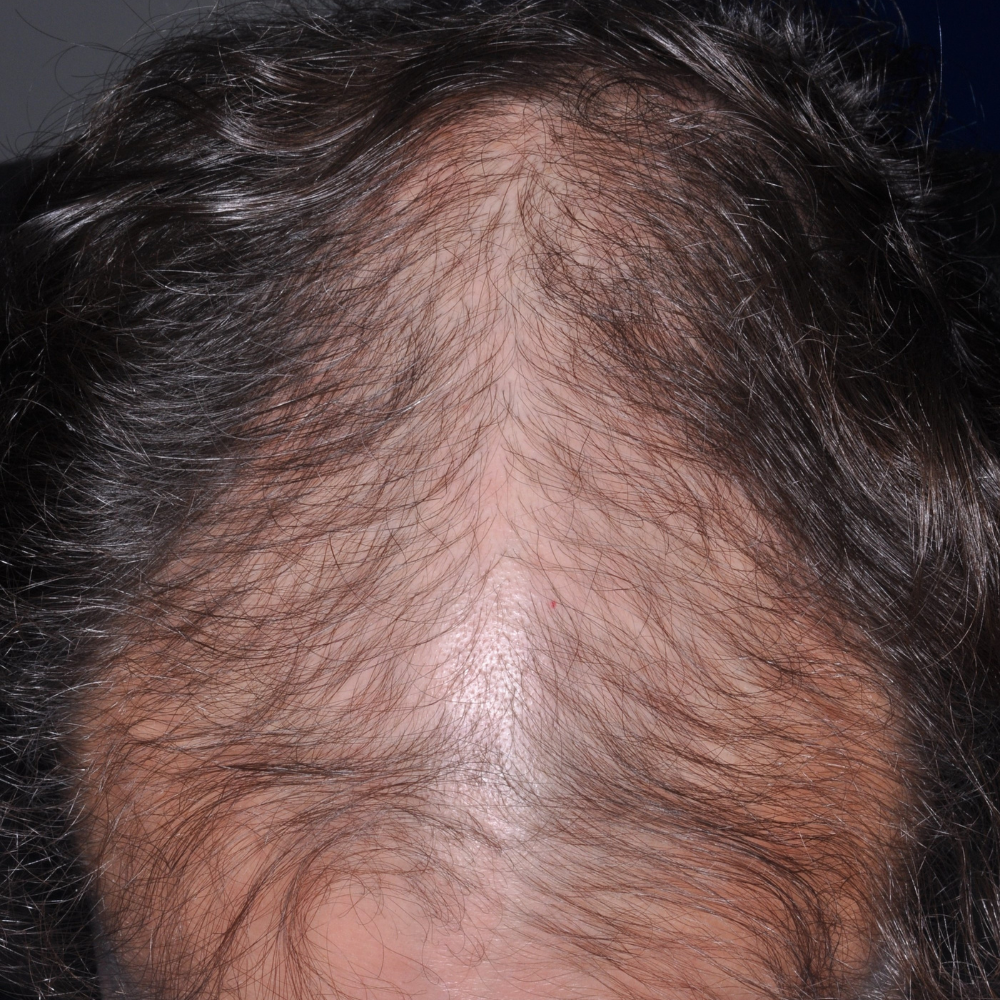
BB Before
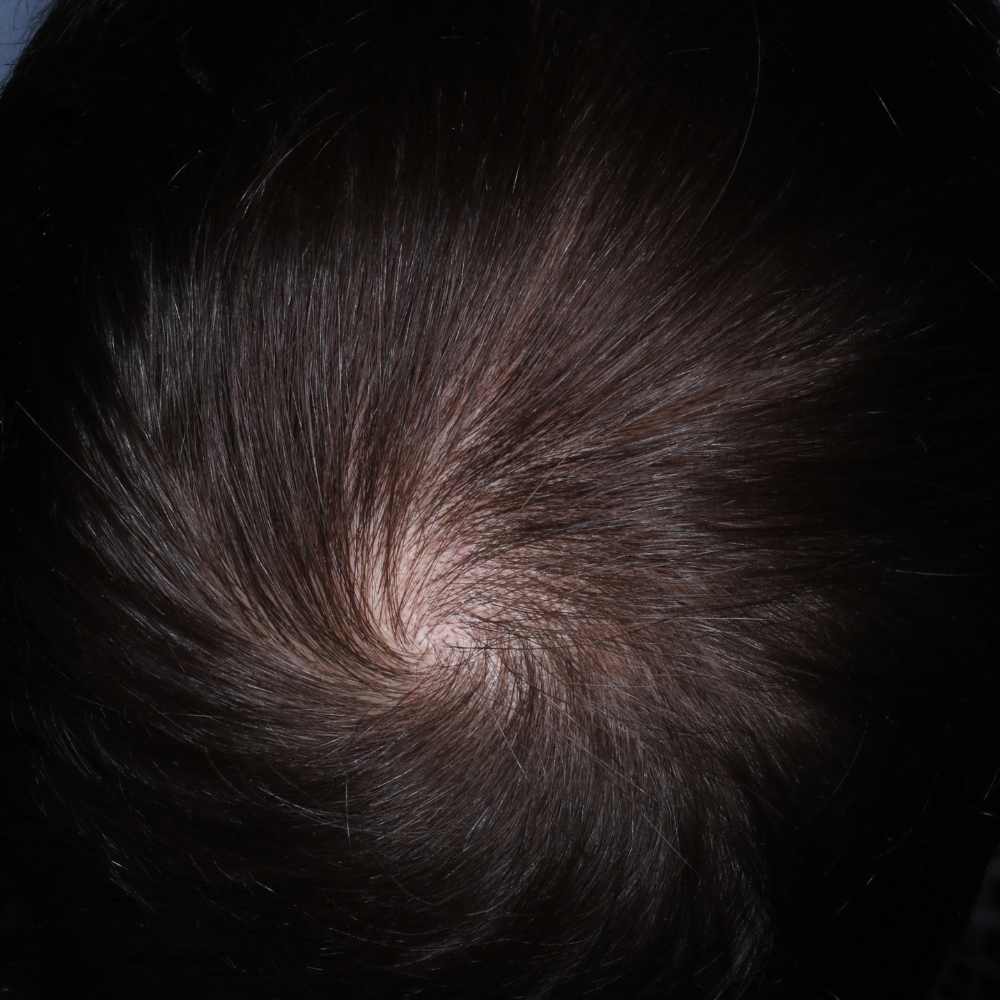
EF After
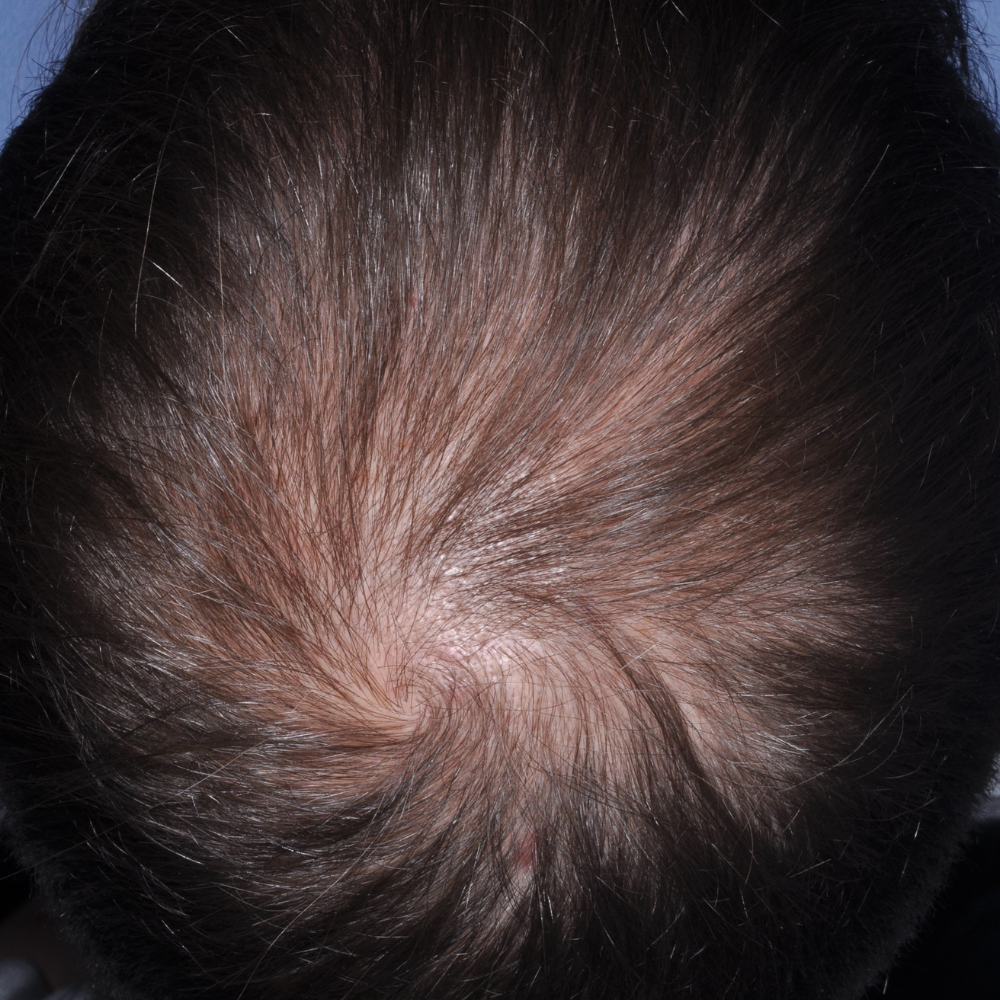
EF Before
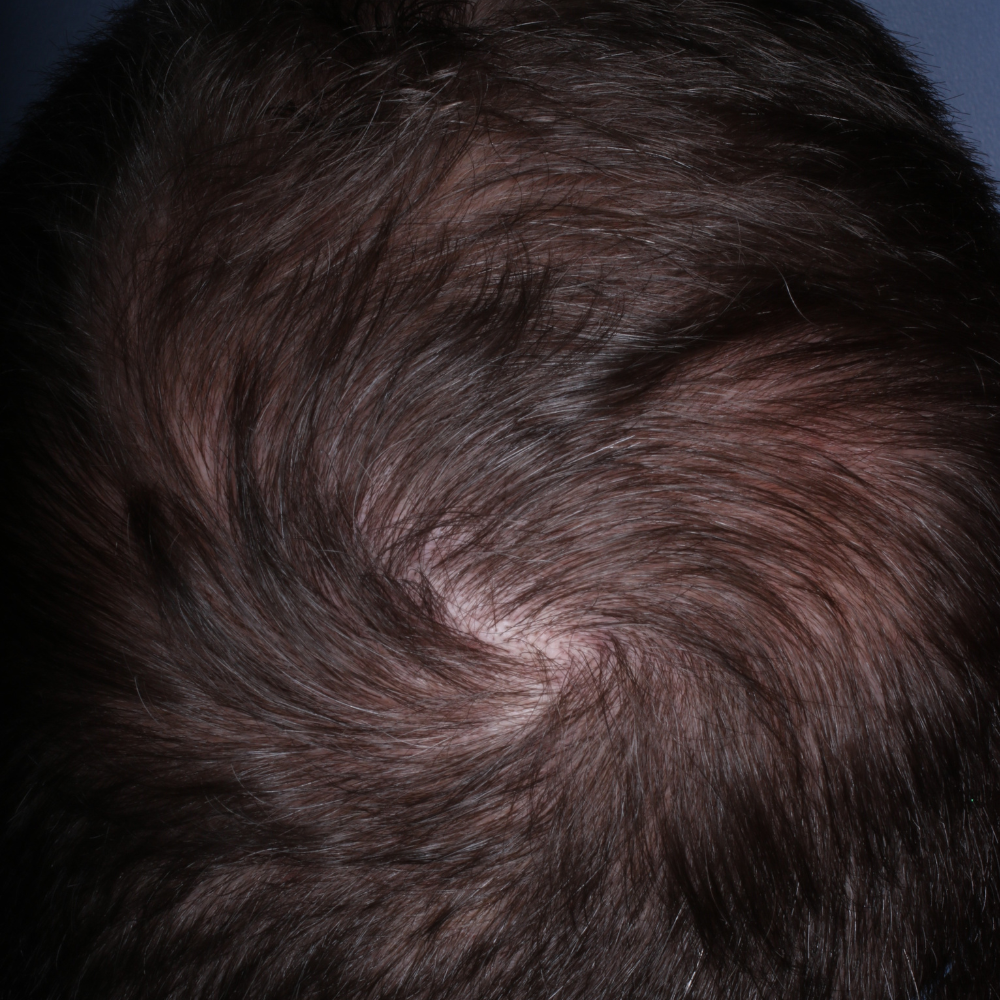
MP After
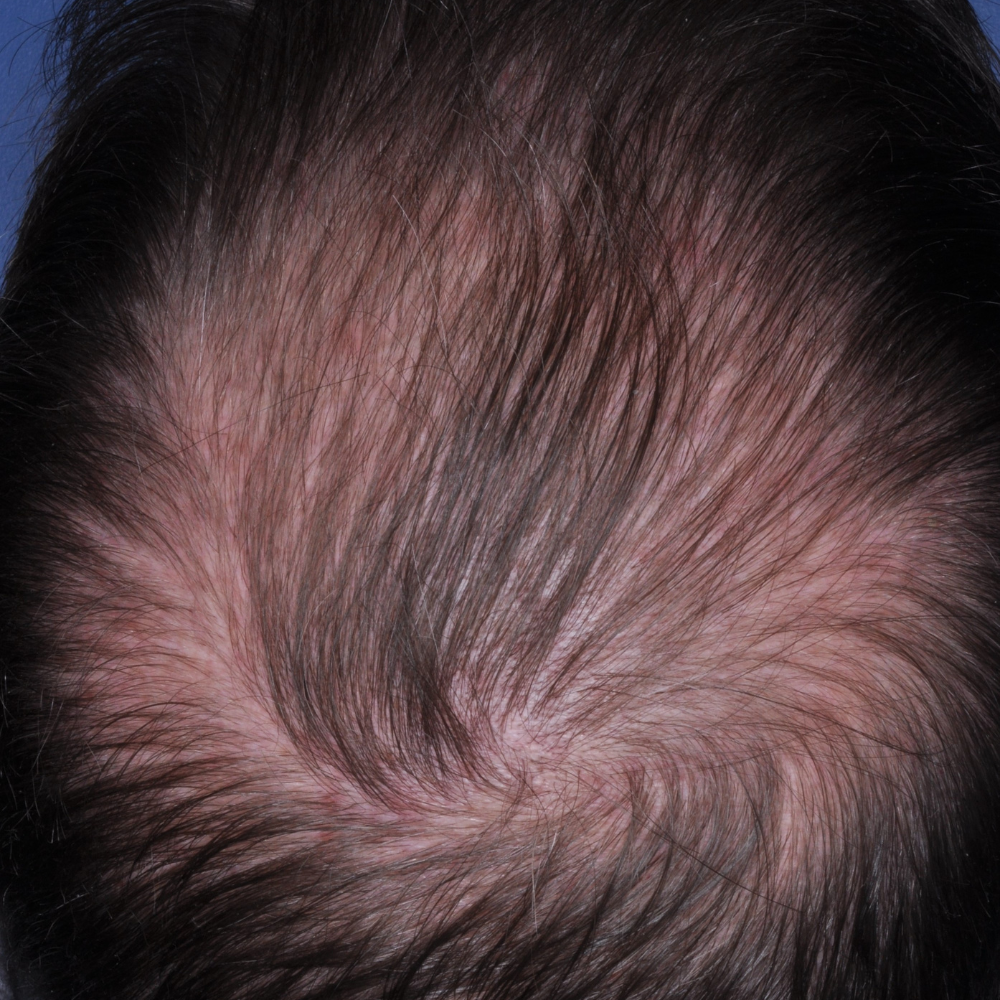
MP Before

MW After

MW Before

SOK After

SOK Before
PCIA

PCIA Before